|
Write out the reactivity series and your mnemonic Which are very reactive and which are unreactive Metal + water -> metal hydroxide + hydrogen Write an example for this equation What are the tests for oxygen, hydrogen and carbon dioxide Corrosion Making Salts Neutralisation Hydrochloric acid = chloride Sulfuric acid = sulfate Nitric acid = nitrate Write examples of making salts using these acids and 3 different metals What does corrosion mean? How could you test to see which conditions allow rusting to take place Iron + oxygen -> Iron oxide Acid + alkali -> salt + water Write an example using the names of acids, alkalis and the salt made. What does this mean? Metal carbonate ->metal oxide + carbon dioxide Write equations for magnesium carbonate, zinc carbonate and copper carbonate when you heat them. Keywords The Reactivity Series Alkali metals in water Displacement reactions Thermal Decomposition Metal oxide + metal -> Metal oxide + metal Find an example in your book to re-write this equation showing a displacement reaction Give examples of words to describe a reaction taking place. Please visit and I hope it will help in your teaching.Presentation on theme: "Write out the reactivity series and your mnemonic"- Presentation transcript:ġ Write out the reactivity series and your mnemonic This is a completely free site and requires no registration. 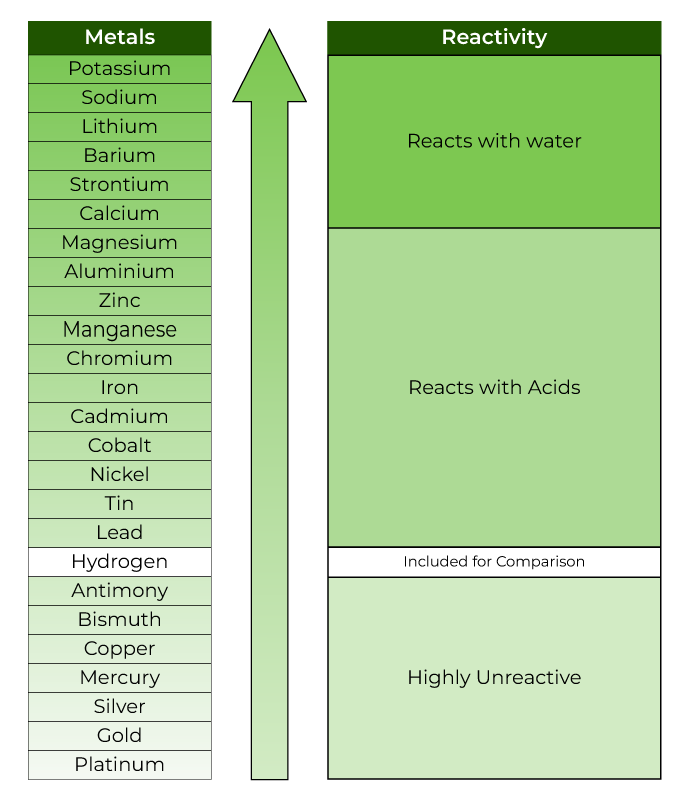 
This powerpoint was kindly donated to is home to over a thousand powerpoints submitted by teachers. The Reactivity Series Increasing reactivity Potassium Sodium Calcium Magnesium Aluminium Carbon Zinc Iron Lead Copper Silver Gold The Reactivity Series lists metals in order of reactivity: Nickel + hydrochloric acid Lithium hydroxide + hydrogen Lithium chloride + hydrogen Silver oxide Magnesium sulphate + hydrogen Potassium oxide Aluminium oxide Manganese oxide + hydrogen Sodium sulphate + hydrogen Lithium oxide Nickel chloride + hydrogen.Lithium + sulphuric acid When a metal reacts with an acid it gives off hydrogen (which can be “popped” using a lit splint).magnesium + hydrochloric acid magnesium chloride + hydrogen Reactions of metals with acids METAL + ACID SALT + HYDROGEN e.g. The other product will be either a metal hydroxide or a metal oxide. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
